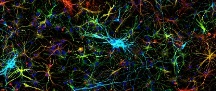
A Multi-Disciplinary Center Developing New Tools and Therapeutics
Neurodegenerative Diseases, Stroke & Traumatic Brain Injury
RNA Binding Proteins and RNA Processing as Targets for Brain Disorders
Research in Assistant Professor of Biomedical Sciences Sika Zheng’s lab is focused on RNA processing and regulation unique to the brain. Through studying brain-specific RNA-centered gene expression mechanisms and the related regulators, i.e., RNA binding proteins, the lab aims to design more refined and targeted approach against various brain disorders without systemic side effects on other parts of the body. The Zheng lab is also actively developing new methods, particularly assays suitable for diagnostics and screening. For example, the lab recently devised a new method to precisely monitor changes in cellular NMD activity, alternation of which could have a large impact on various diseases, including amyotrophic lateral sclerosis, frontotemporal dementia, and cancers. Using these innovative technologies, Zheng and collaborators are performing high to medium throughput molecular screening to identify hit compounds that will be optimized into potential drug leads.
Read more about research into RNA binding proteins and RNA processing
Targeting Remyelination and Neuroprotection in Multiple Sclerosis (MS)
The Tiwari-Woodruff lab investigates mechanisms of demyelination-induced neurodegeneration and therapeutic neuroprotection using two demyelinating mouse models of MS: immune-mediated experimental autoimmune encephalomyelitis (EAE) and non-immune-mediated toxic cuprizone diet. Demyelination related pathologies targeting optic neuritis, learning and memory, seizure activity in MS are some of the topics that are being investigated. Additionally, through continued collaborations with researchers at UIUC, UCLA, and pharmaceutical companies, the lab interrogates remyelinating/therapeutic efficacies and mechanisms of action of various approved and potential MS drugs. As all the estrogen receptor (ER) beta ligands induce axon remyelination and neuroprotection during EAE, we analyzed the common mechanism in otherwise minimal immunomodulation. The ER beta ligands all upregulate the chemokine CXCl1/KC as measured in the peripheral splenocyte during EAE. It is involved in both leukocyte migration and oligodendrocyte development. We are interested in pursuing this target in collaboration with Professor Mauritzio Pellecchia and hope to invent and test small molecules that target CXCl1 increases or CXCR2 receptor activity in the CNS to induce axon remyelination and neuroprotection in multiple sclerosis and other neurodegenerative diseases”.
Read more about research into Multiple Sclerosis
Targeting the EphA4 receptor for the treatment of Amyotrophic Lateral Sclerosis (ALS)
Professor of Biomedical Sciences Maurizio Pellecchia of the School of Medicine and his collaborators have designed a novel series of compounds targeting the EphA4 receptor that are very efficacious in cellular and animal models of ALS. Working in collaboration with the laboratory of Professor of Biomedical Sciences Iryna Ethell, researchers are further studying these agents at the mechanistic level. Iron Horse Therapeutics, a biotech company in San Diego, is attempting to capitalize on this research to develop much needed novel ALS therapeutics.
Fragile X Syndrome (FXS) and Alzheimer’s Disease
Researchers in the laboratory of UCR School of Medicine Professor of Biomedical Sciences Iryna Ethell utilize novel molecular approaches in neuroscience and mouse genetics to conduct research on the molecular basis of neurologic diseases. Through this research, the laboratory is unveiling the mechanisms at the basis to the pathophysiology of Fragile X Syndrome (FXS) and glial control of injury-induced synapse development and remodeling in the brain following traumatic brain injury and diseases; role of actin-regulating proteins in synaptic plasticity, A β-induced synapse loss and cognitive decline associated with Alzheimer's Disease. These studies culminated with the identification of potential drug targets and the development of animal models to assess their efficacy, both essential stepping stones for the development of novel therapeutics.
Read more about Research into Fragile X Syndrome and Alzheimer's Disease
Stroke and Brain Injury
UCR School of Medicine Professor of Biomedical Sciences Byron Ford’s laboratory studies the neuroprotective roles of neuregulin-1 (NRG-1) and other compounds in stroke and other acute neuroinflammatory disorders. Researchers utilize in vivo and in vitro models as well as high-throughput tools to scan the genome, transcriptome, proteome and metabolome to understand mechanisms associated with neuronal pathology and neuroinflammation following acute brain injuries. NRG-1 is currently in human clinical trials and showed significant efficacy in a phase II study of heart failure patients. The results of these studies have therapeutic implications for other acute neuroinflammatory disorders include traumatic brain injury, nerve agent toxicity, cerebral malaria and sepsis.